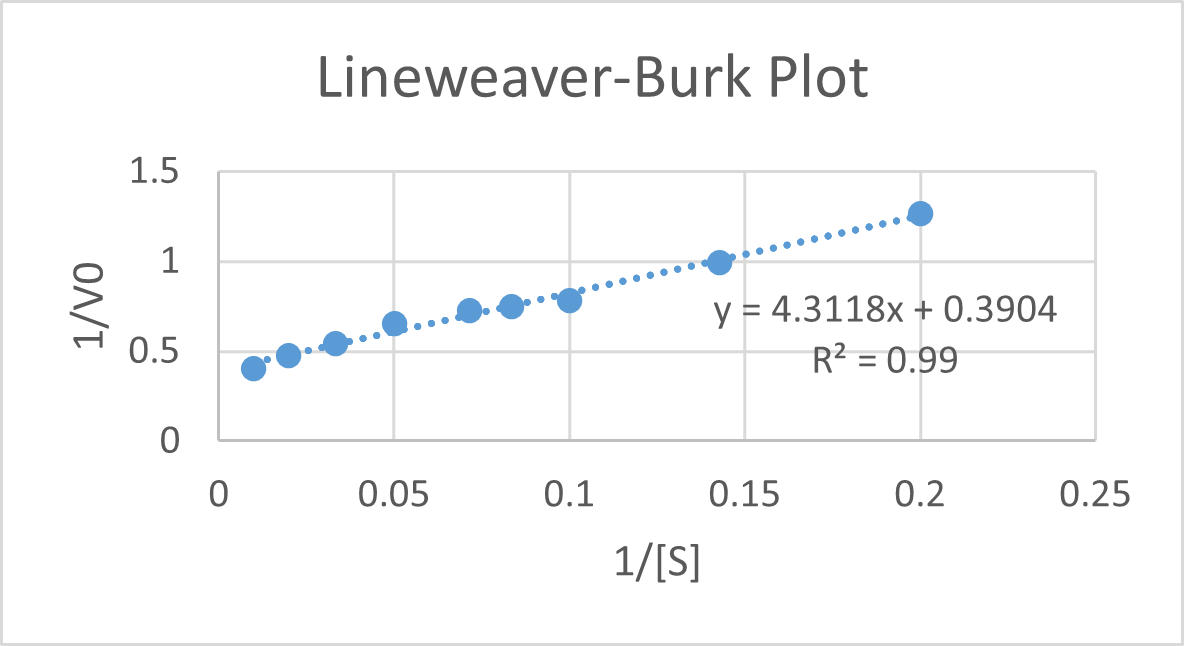
8 (4) A, C 0.1 gm of organic compound was analysed by Kjeldahl's method. In analysis produced NH, absorbed in 30 ml N/5 H,SO. The remaining acid required 20 ml N/10 NaOH
Por um escritor misterioso
Last updated 10 novembro 2024

Click here:point_up_2:to get an answer to your question :writing_hand:84 a c01 gm of organic compound was analysed bykjeldahls method in analysis produced nhabsorbed

Amines.docx

Nitrogen - ScienceDirect

The Kjeldahl Titrimetric Finish: On the Ammonia Titration Trapping in Boric Acid

Reagents used during decomposition methods tested c/(mol L −1 ) Na 2 CO
During estimation of nitrogen present in an organic compound using Kjeldahl's method, the NH3 nbsp;evolved from 0.25 g of the compound was neutralised by 10 ml of 1.25 N H2SO4. What is
21. For the estimation of nitrogen, 1.4 g of an organic compound was digested by Kjeldahl method and the evolved ammonia was absorbed in 60 mL of M/10 sulphuric acid. The unreacted

0.1gm of organic compound was analysed by Kjeldahl's method,In analysi

During nitrogen estimation present in an organic compound by Kje

Methods Manual Soil Testing In India - Department of Agriculture

Kjeldahl Method Questions - Practice Questions of Kjeldahl Method with Answer & Explanations
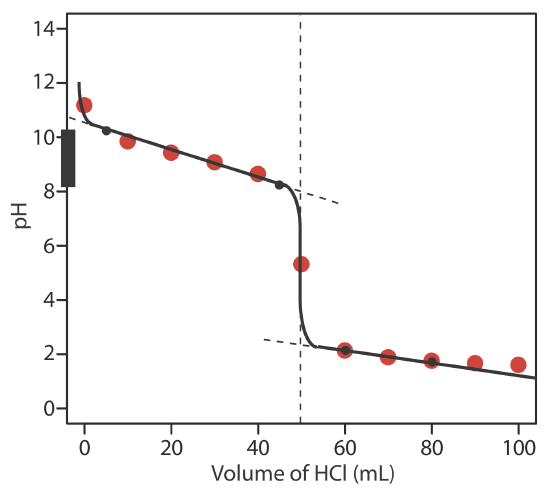
9.2: Acid–Base Titrations - Chemistry LibreTexts

For the estimation of nitrogen, 1.4 g of an organic compound was digested by Kjeldahl's method and
18 29.5 mg of an organic compound containing nitrogen was digested according to Kjeldahls method and the evolved ammonia was absorbed in 20 mL of 0.1 M HCl solution. The excess of

8 (4) A, C 0.1 gm of organic compound was analysed by Kjeldahl's method. In analysis produced NH, absorbed in 30 ml N/5 H,SO. The remaining acid required 20 ml N /10 NaOH

Chemical Engg Reviewer, PDF, Hydroxide
Recomendado para você
-
Analysed databases. Source. Web of Science Core Collection (2022).10 novembro 2024
-
 Analysed synonyms that belongs to adjectives10 novembro 2024
Analysed synonyms that belongs to adjectives10 novembro 2024 -
Solved In Part A you analysed the activity of ADH under10 novembro 2024
-
 Getting My Blood Analysed - How Healthy Am I? - Laura Try10 novembro 2024
Getting My Blood Analysed - How Healthy Am I? - Laura Try10 novembro 2024 -
 Analysed Downloadable Scores10 novembro 2024
Analysed Downloadable Scores10 novembro 2024 -
 12 lead ECG Machine with printer Diagnosis Auto-analysed Result FDA Approved 70072120868510 novembro 2024
12 lead ECG Machine with printer Diagnosis Auto-analysed Result FDA Approved 70072120868510 novembro 2024 -
 Fritjof Capra quote: Whenever the essential nature of things is analysed by the10 novembro 2024
Fritjof Capra quote: Whenever the essential nature of things is analysed by the10 novembro 2024 -
 How much do Tour de France bikes weigh in 2023? 11 Tour bikes weighed and analysed - BikeRadar10 novembro 2024
How much do Tour de France bikes weigh in 2023? 11 Tour bikes weighed and analysed - BikeRadar10 novembro 2024 -
 Sergio Reguilón's stop-start Manchester United career analysed10 novembro 2024
Sergio Reguilón's stop-start Manchester United career analysed10 novembro 2024 -
 We're condemning an innocent woman” - Professor Alvarez, who analysed Simona Halep's hair - Tennis Majors10 novembro 2024
We're condemning an innocent woman” - Professor Alvarez, who analysed Simona Halep's hair - Tennis Majors10 novembro 2024
você pode gostar
-
 Monitor Gamer 24.5 Ozone Dsp25 Ultra - Full Hd - 360hz - Hdr - 0.6ms - G-sync - Freesync - Escorrega o Preço10 novembro 2024
Monitor Gamer 24.5 Ozone Dsp25 Ultra - Full Hd - 360hz - Hdr - 0.6ms - G-sync - Freesync - Escorrega o Preço10 novembro 2024 -
Vk boy band10 novembro 2024
-
 RX6800 16G Game Graphics Card Plates Paca De Video Card Board Gpu Nvidia Geforce PC Computer not 5500 5600 5700 6600 6700 - AliExpress10 novembro 2024
RX6800 16G Game Graphics Card Plates Paca De Video Card Board Gpu Nvidia Geforce PC Computer not 5500 5600 5700 6600 6700 - AliExpress10 novembro 2024 -
 Character Maker (Male)|Picrew10 novembro 2024
Character Maker (Male)|Picrew10 novembro 2024 -
 Bota louis vuitton original - Roupas - Recreio dos Bandeirantes, Rio de Janeiro 123314236510 novembro 2024
Bota louis vuitton original - Roupas - Recreio dos Bandeirantes, Rio de Janeiro 123314236510 novembro 2024 -
Banco Imobiliário Kit Super Jogo de Tabuleiro Clássico Tradicional10 novembro 2024
-
 Os 45 anos de um jogo clássico: o War, da Grow10 novembro 2024
Os 45 anos de um jogo clássico: o War, da Grow10 novembro 2024 -
 base oc/nezuko♡ em 2023 Base de desenho, Desenhando esboços, Base de anime10 novembro 2024
base oc/nezuko♡ em 2023 Base de desenho, Desenhando esboços, Base de anime10 novembro 2024 -
 New Shadow Fruit Showcase10 novembro 2024
New Shadow Fruit Showcase10 novembro 2024 -
 FIDE seeks public discussion about proposed rating changes10 novembro 2024
FIDE seeks public discussion about proposed rating changes10 novembro 2024